A SARS-like virus discovered this summer in the Middle East may infect more than just humans. The pathogen, a close cousin to the one that caused the 2002 to 2003 SARS outbreak, may also be able to infect cells from pigs and a wide range of bat species, researchers report today. The findings may help public health officials track the source of the outbreak and identify the role of wild animals and livestock in spreading the virus, researchers say.
Scientists first detected the virus in a 60-year-old man from Jeddah, Saudi Arabia, who developed severe pneumonia this past spring. Unable to identify the microbe causing the illness, doctors sent samples to Erasmus MC in Rotterdam, the Netherlands. There, scientists identified the infectious agent as a coronavirus, a group known to cause many ailments, such as the common cold and a variety of gastrointestinal infections. Cases have popped up in Qatar and Jordan as well; in total, researchers have so far confirmed nine infections, including five deaths. Several other cases are suspected but haven’t been confirmed.
Researchers have fully sequenced the virus, which they dubbed hCoV-EMC (short for human coronavirus-Erasmus Medical Center). The genome revealed that it is closely related to the SARS coronavirus.
The new study, published online in mBio, is an attempt to answer other basic questions, such as where the virus originated, how it enters cells, and what other animals it might infect, says Christian Drosten, a virologist at the University of Bonn Medical Center in Germany and one of the lead authors.
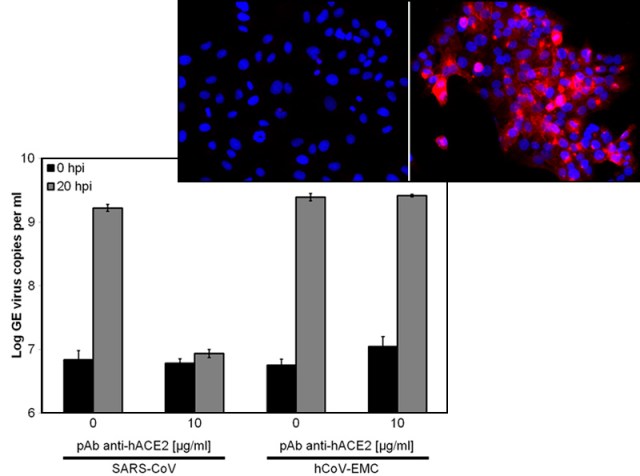
Scientists knew that the SARS virus uses a receptor called ACE2 to pry open cells. Because these receptors are mainly found deep inside the human lung, patients developed very severe illness that frequently left them too sick to spread SARS to many others; the people most at risk were health care workers who take care of patients. If hCoV-EMC used the same receptor, researchers would have a head start in understanding how it spreads and how to stop it—primarily by protecting health care workers. It might also help them in the development of drugs and vaccines.
To find out, the team engineered baby hamster kidney cells to express the human ACE2 receptor. These cells could be infected with the SARS coronavirus, as expected, but not hCoV-EMC. That finding, supported by additional experiments, led them to conclude that the new coronavirus does not use ACE2 to get in. Which receptor it uses instead is still unclear, which is a “downside” of the new study, says Larry Anderson, an infectious disease specialist at Emory University in Atlanta.
Epidemiologists also want to know which species of animals it is capable of infecting to keep the new coronavirus from spreading further. To determine what types of animals hCoV-EMC can infect, Drosten and colleagues infected cells from humans, pigs, and a wide variety of bats, the key natural reservoirs of coronaviruses. The new virus could infect all of these types of cells. “It’s unusual for a coronavirus to easily go back to bats,” Drosten says. “Most coronaviruses come from bats, but once they jump to other species, you could never get them to reinfect bat cells.” The SARS virus, for instance, originated in Chinese horseshoe bats, but once it ended up in humans, it had changed so much that scientists were unable to infect bat cells with it.
“The fact that [hCoV-EMC] can infect bat cells is consistent with the hypothesis that bats might be the origin of this virus, but this finding doesn’t prove it,” Anderson says. “This virus had to come from an animal source—there’s no other explanation for what’s going on. But we still don’t know what that source is.”
Based on the findings, however, it seems likely that the new coronavirus can infect a wide range of species, Drosten says. That means public health officials may have to start looking for infections and deaths in local wild animal and livestock populations to keep the virus in check, he says.
http://news.sciencemag.org/sciencenow/2012/12/new-sars-like-virus-infects-both.html?ref=hp