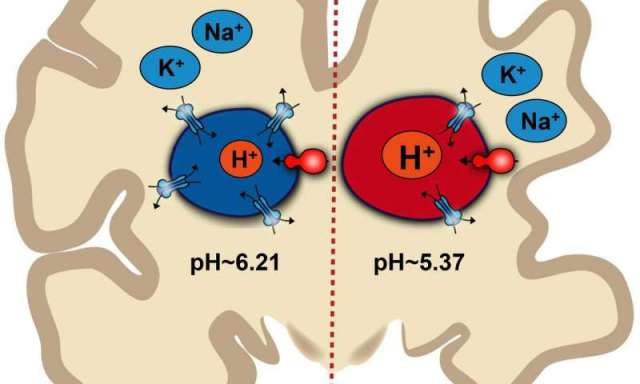
Illustration of how pH imbalance inside endosomes may contribute to Alzheimer’s disease
Johns Hopkins Medicine scientists say they have found new evidence in lab-grown mouse brain cells, called astrocytes, that one root of Alzheimer’s disease may be a simple imbalance in acid-alkaline—or pH—chemistry inside endosomes, the nutrient and chemical cargo shuttles in cells.
Astrocytes work to clear so-called amyloid beta proteins from the spaces between neurons, but decades of evidence has shown that if the clearing process goes awry, amyloid proteins pile up around neurons, leading to the characteristic amyloid plaques and nerve cell degeneration that are the hallmarks of memory-destroying Alzheimer’s disease.
The new study, described online June 26 in Proceedings of the National Academy of Sciences, also reports that the scientists gave drugs called histone deacetylase (HDAC) inhibitors to pH-imbalanced mice cells engineered with a common Alzheimer’s gene variant. The experiment successfully reversed the pH problem and improved the capacity for amyloid beta clearance.
HDAC inhibitors are approved by the U.S. Food and Drug Administration for use in people with certain types of blood cancers, but not in people with Alzheimer’s. They cautioned that most HDAC inhibitors cannot cross the blood-brain barrier, a significant challenge to the direct use of the drugs for brain disorders. The scientists say they are planning additional experiments to see if HDAC inhibitors have a similar effect in lab-grown astrocytes from Alzheimer’s patients, and that there is the potential to design HDAC inhibitors that can cross the barrier.
However, the scientists caution that even before those experiments can happen, far more research is needed to verify and explain the precise relationship between amyloid proteins and Alzheimer’s disease, which affects an estimated 50 million people worldwide. To date, there is no cure and no drugs that can predictably or demonstrably prevent or reverse Alzheimer’s disease symptoms.
“By the time Alzheimer’s disease is diagnosed, most of the neurological damage is done, and it’s likely too late to reverse the disease’s progression,” says Rajini Rao, Ph.D., professor of physiology at the Johns Hopkins University School of Medicine. “That’s why we need to focus on the earliest pathological symptoms or markers of Alzheimer’s disease, and we know that the biology and chemistry of endosomes is an important factor long before cognitive decline sets in.”
Nearly 20 years ago, scientists at Johns Hopkins and New York University discovered that endosomes, circular compartments that ferry cargo within cells, are larger and far more abundant in brain cells of people destined to develop Alzheimer’s disease. This hinted at an underlying problem with endosomes that could lead to an accumulation of amyloid protein in spaces around neurons, says Rao.
To shuttle their cargo from place to place, endosomes use chaperones—proteins that bind to specific cargo and bring them back and forth from the cell’s surface. Whether and how well this binding occurs depends on the proper pH level inside the endosome, a delicate balance of acidity and alkalinity, or acid and base, that makes endosomes float to the surface and slip back down into the cell.
Embedded in the endosome membrane are proteins that shuttle charged hydrogen atoms, known as protons, in and out of endosomes. The amount of protons inside the endosome determines its pH.
When fluids in the endosome become too acidic, the cargo is trapped within the endosome deep inside the cell. When the endosome contents are more alkaline, the cargo lingers at the cell’s surface for too long.
To help determine whether such pH imbalances occur in Alzheimer’s disease, Johns Hopkins graduate student Hari Prasad scoured scientific studies of Alzheimer’s disease looking for genes that were dialed down in diseased brains compared with normal ones. Comparing a dataset of 15 brains of Alzheimer’s disease patients with 12 normal ones, he found that 10 of the 100 most frequently down-regulated genes were related to the proton flow in the cell.
In another set of brain tissue samples from 96 people with Alzheimer’s disease and 82 without it, gene expression of the proton shuttle in endosomes, known as NHE6, was approximately 50 percent lower in people with Alzheimer’s disease compared with those with normal brains. In cells grown from people with Alzheimer’s disease and in mouse astrocytes engineered to carry a human Alzheimer’s disease gene variant, the amount of NHE6 was about half the amount found in normal cells.
To measure the pH balance within endosomes without breaking open the astrocyte, Prasad and Rao used pH sensitive probes that are absorbed by endosomes and emit light based on pH levels. They found that mouse cell lines containing the Alzheimer’s disease gene variant had more acidic endosomes (average of 5.37 pH) than cell lines without the gene variant (average of 6.21 pH).
“Without properly functioning NHE6, endosomes become too acidic and linger inside astrocytes, avoiding their duties to clear amyloid beta proteins,” says Rao.
While it’s likely that changes in NHE6 happen over time in people who develop sporadic Alzheimer’s disease, people who have inherited mutations in NHE6 develop what’s known as Christianson syndrome in infancy and have rapid brain degeneration.
Prasad and Rao also found that a protein called LRP1, which picks up amyloid beta proteins outside the astrocyte and delivers them to endosomes, was half as abundant on the surface of lab grown mouse astrocytes engineered with a human gene variant called APOE4, commonly linked to Alzheimer’s disease.
Looking for ways to restore the function of NHE6, Prasad searched databases of yeast studies to find that HDAC inhibitors tend to increase expression of the NHE6 gene in yeast. This gene is very similar across species, including flies, mice and humans.
Prasad and Rao tested nine types of HDAC inhibitors on cell cultures of mouse astrocytes engineered with the APOE4 gene variant. Broad-spectrum HDAC inhibitors increased NHE6 expression to levels associated with mouse astrocytes that did not have the Alzheimer’s gene variant. They also found that HDAC inhibitors corrected the pH imbalance inside endosomes and restored LRP1 to the astrocyte surface, resulting in efficient clearance of amyloid beta protein.
More information: Hari Prasad et al. Amyloid clearance defect in ApoE4 astrocytes is reversed by epigenetic correction of endosomal pH, Proceedings of the National Academy of Sciences (2018). DOI: 10.1073/pnas.1801612115
https://medicalxpress.com/news/2018-08-ph-imbalance-brain-cells-contribute.html