The Food and Drug Administration (FDA) approved Friday the first medical device to treat childhood attention deficit hyperactivity disorder, or ADHD.
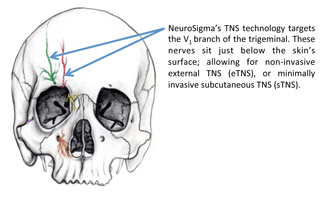
The device, approved for children ages 7 to 12 who do not currently take ADHD medicine, delivers a low-level electrical pulse to a patch placed on the forehead that interacts with the parts of the brain responsible for ADHD symptoms.
“This new device offers a safe, non-drug option for treatment of ADHD in pediatric patients through the use of mild nerve stimulation, a first of its kind,” Carlos Peña, director of the Division of Neurological and Physical Medicine Devices in the FDA’s Center for Devices and Radiological Health, said in a statement.
“Today’s action reflects our deep commitment to working with device manufacturers to advance the development of pediatric medical devices so that children have access to innovative, safe and effective medical devices that meet their unique needs.”
The device, called the Monarch external Trigeminal Nerve Stimulation System (eTNS), is marketed by NeuroSigma and is only available by prescription and must be monitored by a caregiver.
eTNS, which is designed to fit inside one’s pocket, is connected by wire to a patch that is placed on the forehead while sleeping and delivers a “tingling” electrical impulse to branches of the trigeminal nerve.
“While the exact mechanism of eTNS is not yet known, neuroimaging studies have shown that eTNS increases activity in the brain regions that are known to be important in regulating attention, emotion and behavior,” the FDA said.
The FDA also conducted a trial with 62 children with ADHD in which a group that used eTNS had “significant improvement” in their symptoms as opposed to another group that used a placebo.
Side effects of the treatment include drowsiness, an increase in appetite, trouble sleeping, teeth clenching, headache and fatigue. The device should not be placed near a phone or used by children on an insulin pump, pacemaker or implanted neurostimulator, the FDA said.
The FDA found no serious adverse events associated with eTNS.
The device is not currently covered by insurance and has a price tag of just over $1,000 for a starter kit, according to the NeuroSigma website.
FDA gives green light to first medical device to treat ADHD in children


