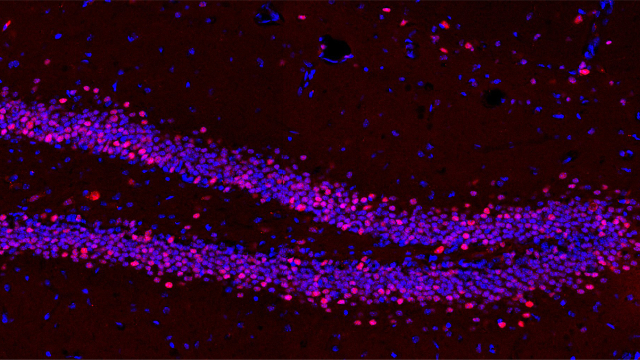
The dentate gyrus of a mouse that received deep brain stimulation, with cell nuclei in blue and expression of the gene c-Fos in red.
By Shawna Williams
Even as patients with Parkinson’s disease, obsessive-compulsive disorder, and other conditions turn to deep brain stimulation (DBS) to keep their symptoms in check, it’s been unclear to scientists why the therapy works. Now, researchers in Texas report that in mice, the treatment dials the activity of hundreds of genes up or down in brain cells. Their results, published in eLife March 23, hint that DBS’s use could be expanded to include improving learning and memory in people with intellectual disabilities.
“The paper is very well done. . . . It’s really a rigorous study,” says Zhaolan “Joe” Zhou, a neuroscientist at the University of Pennsylvania’s Perelman School of Medicine who reviewed the paper for eLife. Now that the genes and pathways DBS affects are known, researchers can home in on ways to improve the treatment, or perhaps combine the therapy with pharmacological approaches to boost its effect, he says.
In DBS, two electrodes are surgically implanted in a patient’s brain (the area depends on the disorder being treated), and connected to generators that are placed in the chest. Gentle pulses of electricity are then passed continuously through the electrodes. The treatment reduces motor symptoms in many people with Parkinson’s, and allows some patients to reduce their use of medications, but it does not eliminate symptoms or slow the disease’s progression.
In addition to its use in movement disorders, DBS is being explored as a potential therapy for a range of other brain-related disorders. For instance, as a way to boost learning and memory in people with Alzheimer’s disease, researchers are looking into stimulating the fimbria-fornix, a brain region thought to regulate the activity of the memory-storing hippocampus.
Such studies made Huda Zoghbi, a neurogeneticist at Baylor College of Medicine, wonder what effect DBS might have on learning- and memory-related disorders that strike earlier in life. “We rationalized that maybe in Alzheimer’s, many of the neurons are already gone, but perhaps in a healthier brain, like that of a Rett syndrome model, we can test the idea if stimulation of the fornix can improve learning and memory,” she explains. Rett syndrome, a genetic disease that almost exclusively strikes girls, includes intellectual disability, autism-like deficits in social interactions, and a loss of motor function. Several years ago, Zoghbi and colleagues tried zapping the fimbria-fornix, a C-shape bundle of nerves adjacent to the hippocampus in the brain, in mouse models of Rett syndrome. Published in 2015, their results showed that after two weeks of daily, one-hour DBS sessions, the mice with an intellectual disability performed like their peers without the disorder on a range of hippocampus-dependent tasks.
“We were struck that everything became indistinguishable after deep brain stimulation from a baseline normal,” Zoghbi says. This prompted her team to ask, “How does it work at a molecular level?” The answer, she thought, could determine whether DBS of the fimbria-fornix has the potential to serve as a multipurpose tool, treating not just Rett syndrome but other childhood-onset intellectual disabilities with a variety of causes. “It’s going to be really tough, perhaps, to solve these diseases one gene at a time, so that learning can be corrected,” she says. “You could eventually consider an intervention that can be broadly applicable, irrespective of the molecular cause of the defect.”
For the latest study, the research team analyzed baseline differences in gene activity between mice with and without the Rett syndrome–like condition in a part of the hippocampus called the dentate gyrus. They also treated the mice with the intellectual disability once with 45 minutes of DBS. Of the many genes with marked differences in initial activity between the two groups of mice, one-quarter (39 genes) became normal in the Rett mice after treatment, they report.
Zoghbi’s group also tested the effects of DBS in normal mice; in addition to changing the activity levels of thousands of genes, the researchers found, the treatment prompted alternative splicing of the RNA copies of other genes, which would result in differences in the resulting proteins. Many of the genes affected by the alternative splicing are known to be involved in the growth of new neurons or in maintaining the synapses through which brain cells communicate. In the 2015 study, the group had found that DBS enhances some hippocampus-related abilities in wildtype mice, such as spatial learning.
For hints as to whether DBS might have the potential to treat intellectual disabilities other than Rett syndrome, the researchers compared their list of genes whose activity levels changed after DBS in normal mice with existing data on genes known to have abnormal expression levels in mouse models of several such disorders. As with Rett syndrome, DBS in wildtype mice altered the activity levels of about one-quarter of the genes involved in each of the disorders.
The fact that a short period of stimulation had such profound effects on gene expression is interesting, says Svjetlana Miocinovic, a movement-disorders neurologist at Emory University who was not involved in the study. Most research on the mechanism of DBS has focused on changes it induces in the electrical or physical properties of the brain, she tells The Scientist. “I think this kind of study, where they actually look at the molecular environment in these neurons that are exposed to stimulation . . . is really the way to figure out what exactly is going on and how is that neural plasticity accomplished.”
Now that they have a way to measure such molecular effects, Zoghbi and her collaborators plan to optimize DBS for models of intellectual disabilities—figuring out how long the current needs to be on, for example, and how often. Another question they’d like to address is whether stimulating other brain areas in addition to the fimbria-fornix could add to the benefits seen in the mice.
Zoghbi emphasizes that even if DBS turns out to be safe and effective for children with Rett syndrome, it won’t be a silver bullet, because patients will have missed out on some important developmental milestones. “To really get the full benefit,” she says, “we’re going to have to combine any intervention with intensive physical and behavioral therapy.”
A. Pohodish et al., “Forniceal deep brain stimulation induces gene expression and splicing changes that promote neurogenesis and plasticity,” eLife, doi:10.7554/eLife.34031, 2018.