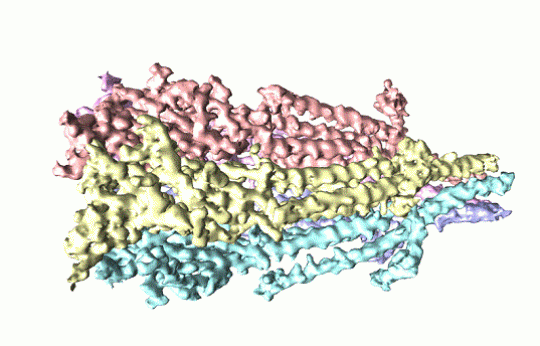
3D reconstruction of a serotonin receptor generated by cryo-electron microscopy
by Rebecca Pool
Claiming a world first and using cryo-electron microscopy, researchers from Case Western Reserve University School of Medicine, US, have observed full-length serotonin receptors. The proteins are common drug targets, and the new images provide details about molecular binding sites that could lead to more precise drug design. Serotonin receptors, which reside in cell membranes throughout the body, are highly dynamic and difficult to image. In the past, the receptors have been sectioned into pieces to study, but by capturing full-length samples, researchers have revealed how different portions interact.
Dr Sandip Basak from Physiology and Biophysics, and colleagues, describe ‘a finely tuned orchestration of three domain movements’ that allows the receptors to elegantly control passageways across cell membranes. “The serotonin receptor acts as a gateway, or channel, from outside the cell to inside,” he says. “When serotonin binds onto the receptor, the channel switches conformation from closed to open. It eventually twists into a ‘desensitized’ state, where the channel closes but serotonin remains attached,” he adds. “This prevents it from being reactivated.”
For this study, the researchers used a FEI Titan Krios microscope, operating at 300 kV, and equipped with a Gatan K2-Summit direct detector camera, at the National Cryo-Electron Microscopy Facility in Frederick, Maryland.
“Successful design of safer therapeutics [for cancer therapies and gastrointestinal diseases] has slowed because there is currently a limited understanding of the structure of the serotonin receptor itself, and what happens after serotonin binds,” says research leader, Professor Sudha Chakrapani. “Our new structure of the serotonin receptor in the resting state has the potential to serve as a structural blueprint to drive targeted drug design and better therapeutic strategies.”
This research is published in Nature Communications.
