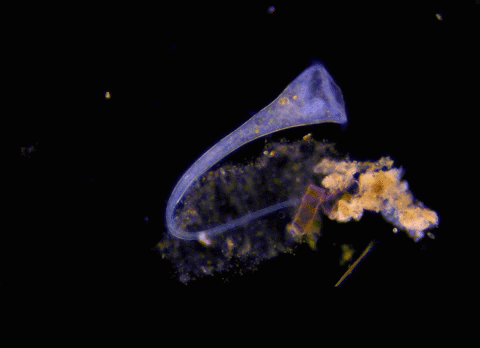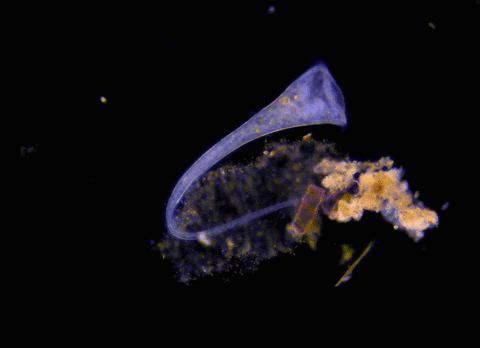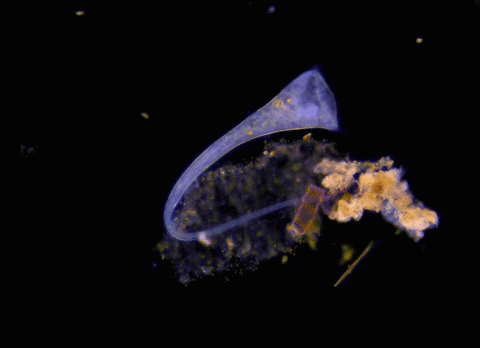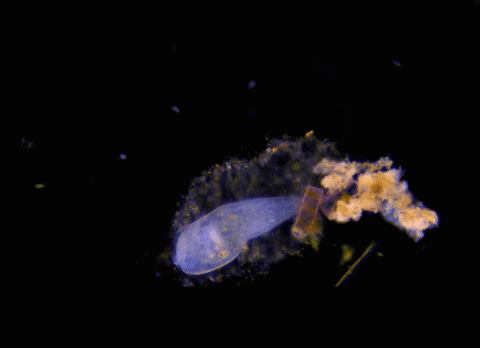
Pinpoint stimulation of a cluster of nerve cells in the brains of mice encouraged timid responses to a perceived threat, whereas stimulation of an adjacent cluster induced boldness and courage.
Researchers at the Stanford University School of Medicine have identified two adjacent clusters of nerve cells in the brains of mice whose activity level upon sighting a visual threat spells the difference between a timid response and a bold or even fierce one.
Located smack-dab in the middle of the brain, these clusters, or nuclei, each send signals to a different area of the brain, igniting opposite behaviors in the face of a visual threat. By selectively altering the activation levels of the two nuclei, the investigators could dispose the mice to freeze or duck into a hiding space, or to aggressively stand their ground, when approached by a simulated predator.
People’s brains probably possess equivalent circuitry, said Andrew Huberman, PhD, associate professor of neurobiology and of ophthalmology. So, finding ways to noninvasively shift the balance between the signaling strengths of the two nuclei in advance of, or in the midst of, situations that people perceive as threatening may help people with excessive anxiety, phobias or post-traumatic stress disorder lead more normal lives.
“This opens the door to future work on how to shift us from paralysis and fear to being able to confront challenges in ways that make our lives better,” said Huberman, the senior author of a paper describing the experimental results. It was published online May 2 in Nature. Graduate student Lindsey Salay is the lead author.
Perilous life of a mouse
There are plenty of real threats in a mouse’s world, and the rodents have evolved to deal with those threats as best they can. For example, they’re innately afraid of aerial predators, such as a hawk or owl swooping down on them. When a mouse in an open field perceives a raptor overhead, it must make a split-second decision to either freeze, making it harder for the predator to detect; duck into a shelter, if one is available; or to run for its life.
To learn how brain activity changes in the face of such a visual threat, Salay simulated a looming predator’s approach using a scenario devised some years ago by neurobiologist Melis Yilmaz Balban, PhD, now a postdoctoral scholar in Huberman’s lab. It involves a chamber about the size of a 20-gallon fish tank, with a video screen covering most of its ceiling. This overhead screen can display an expanding black disc simulating a bird-of-prey’s aerial approach.
Looking for brain regions that were more active in mice exposed to this “looming predator” than in unexposed mice, Salay pinpointed a structure called the ventral midline thalamus, or vMT.
Salay mapped the inputs and outputs of the vMT and found that it receives sensory signals and inputs from regions of the brain that register internal brain states, such as arousal levels. But in contrast to the broad inputs the vMT receives, its output destination points were remarkably selective. The scientists traced these outputs to two main destinations: the basolateral amygdala and the medial prefrontal cortex. Previous work has tied the amygdala to the processing of threat detection and fear, and the medial prefrontal cortex is associated with high-level executive functions and anxiety.
Further inquiry revealed that the nerve tract leading to the basolateral amygdala emanates from a nerve-cell cluster in the vMT called the xiphoid nucleus. The tract that leads to the medial prefrontal cortex, the investigators learned, comes from a cluster called the nucleus reuniens, which snugly envelopes the xiphoid nucleus.
Next, the investigators selectively modified specific sets of nerve cells in mice’s brains so they could stimulate or inhibit signaling in these two nerve tracts. Exclusively stimulating xiphoid activity markedly increased mice’s propensity to freeze in place in the presence of a perceived aerial predator. Exclusively boosting activity in the tract running from the nucleus reuniens to the medial prefrontal cortex in mice exposed to the looming-predator stimulus radically increased a response seldom seen under similar conditions in the wild or in previous open-field experiments: The mice stood their ground, right out in the open, and rattled their tails, an action ordinarily associated with aggression in the species.
Thumping tails
This “courageous” behavior was unmistakable, and loud, Huberman said. “You could hear their tails thumping against the side of the chamber. It’s the mouse equivalent of slapping and beating your chest and saying, ‘OK, let’s fight!’” The mice in which the nucleus reuniens was stimulated also ran around more in the chamber’s open area, as opposed to simply running toward hiding places. But it wasn’t because nucleus reuniens stimulation put ants in their pants; in the absence of a simulated looming predator, the same mice just chilled out.
In another experiment, the researchers showed that stimulating mice’s nucleus reuniens for 30 seconds before displaying the “looming predator” induced the same increase in tail rattling and running around in the unprotected part of the chamber as did vMT stimulation executed concurrently with the display. This suggests, Huberman said, that stimulating nerve cells leading from the nucleus reunions to the prefrontal cortex induces a shift in the brain’s internal state, predisposing mice to act more boldly.
Another experiment pinpointed the likely nature of that internal-state shift: arousal of the autonomic nervous system, which kick-starts the fight, flight or freeze response. Stimulating either the vMT as a whole or just the nucleus reuniens increased the mice’s pupil diameter — a good proxy of autonomic arousal.
On repeated exposures to the looming-predator mockup, the mice became habituated. Their spontaneous vMT firing diminished, as did their behavioral responses. This correlates with lowered autonomic arousal levels.
Human brains harbor a structure equivalent to the vMT, Huberman said. He speculated that in people with phobias, constant anxiety or PTSD, malfunctioning circuitry or traumatic episodes may prevent vMT signaling from dropping off with repeated exposure to a stress-inducing situation. In other experiments, his group is now exploring the efficacy of techniques, such as deep breathing and relaxation of visual fixation, in adjusting the arousal states of people suffering from these problems. The thinking is that reducing vMT signaling in such individuals, or altering the balance of signaling strength from their human equivalents of the xiphoid nucleus and nucleus reuniens may increase their flexibility in coping with stress.
Reference:
Salay, L. D., Ishiko, N., & Huberman, A. D. (2018). A midline thalamic circuit determines reactions to visual threat. Nature. doi:10.1038/s41586-018-0078-2
http://med.stanford.edu/news/all-news/2018/05/scientists-find-fear-courage-switches-in-brain.html